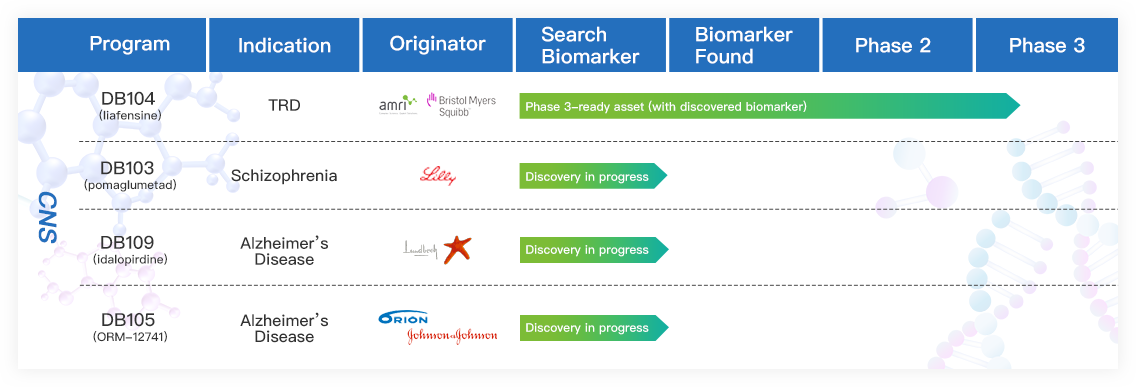
Denovo Neuroscience is revolutionizing psychiatric treatment by developing precision medicines for CNS diseases. It has four first-in-class CNS programs with the lead asset, liafensine, a biomarker guided antidepressant, in phase 3 development in treatment-resistant depression (TRD).
Liafensine is a first–in–class triple reuptake inhibitor that simultaneously targets the serotonin, norepinephrine and dopamine transporters. Through its proprietary, big-data–enabled DGM™ biomarker discovery platform, Denovo identified a novel genetic biomarker in the ANK3 gene that predicts liafensine's efficacy in patients with TRD. In a prospective, biomarker-guided global phase 2b study, liafensine demonstrated robust efficacies in treating ANK3 biomarker-positive patients with TRD - one of the toughest indications to treat.
This work represents the first successful demonstration of precision medicine in psychiatry and validates Denovo’s innovative strategy of rescuing and advancing previously discontinued medicines through biomarker-driven development.